Most of the world’s energy demands are still being met by burning fossil fuels, thereby releasing carbon dioxide into the atmosphere. The energy system is in the midst of a transition to renewable sources such as solar and wind power, but it will be quite some time before fossil fuels are only a minor part of energy production. To reduce global warming, it will be necessary to prevent the carbon dioxide from fossil fuels from entering the atmosphere by capturing it and either locking it away or making use of it.
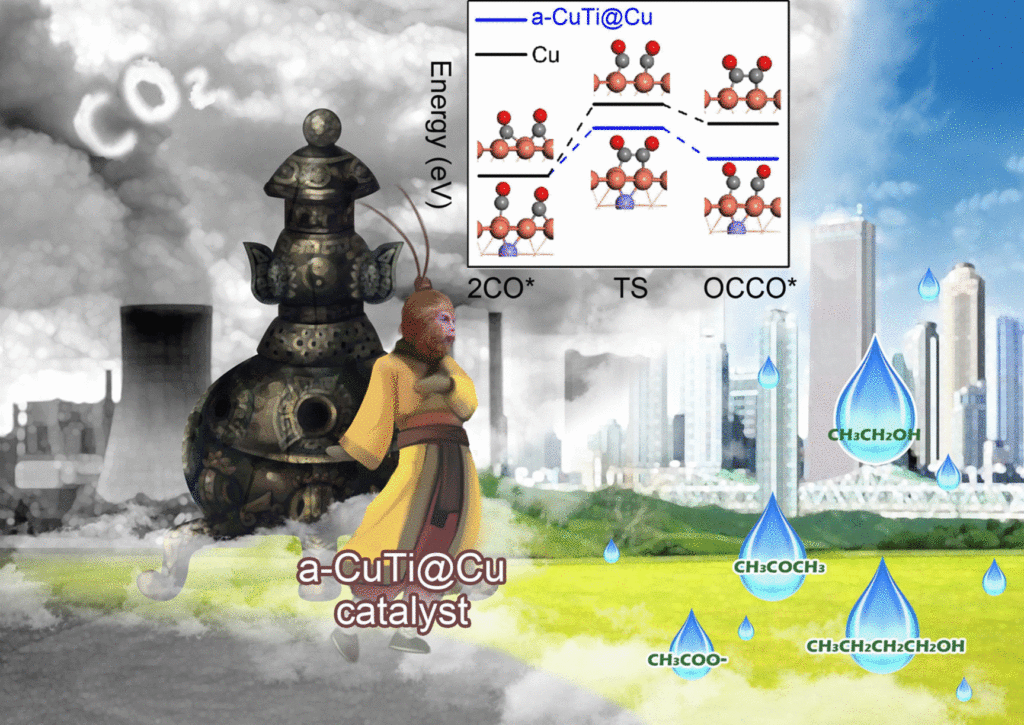
Recent research at several Chinese universities has developed a novel electrocatalyst that efficiently converts CO2 to liquid fuels containing multiple carbon atoms. The main products of the high-efficiency reaction are ethanol, acetone, and n-butanol. Previous electrocatalystic methods have mostly produced simpler hydrocarbons – namely, ones with only a single carbon atom. The fuels the new catalyst produces are much more useful.
The catalyst is made from thin ribbons of a copper/titanium alloy that are etched with hydrofluoric acid to remove the titanium from the surface. The process results in a material with a porous copper surface on an amorphous CuTi alloy. The substance exhibits remarkably high activity, selectivity, and stability for catalyzing the reactions leading to the production of the hydrocarbon fuels.
Converting carbon dioxide into liquid fuels would be advantageous because they have high energy density and are safe to store and transport. Apart from preventing carbon dioxide from entering the atmosphere, the process could also be a way to make use of excess energy produced by solar and wind generation by essentially storing that energy in the form of liquid fuels.
**********
Web Links
Liquid Fuels from Carbon Dioxide
Photo courtesy of Angewandte Chemie via Wiley-VCH.
Earth Wise is a production of WAMC Northeast Public Radio.
Leave a Reply